Electrical resistance
From Wikipedia, the free encyclopedia
A 750-kΩ resistor, as
identified by its electronic color code. An ohmmeter could
be used to verify this value.
Electrical resistance is a measure of the degree to which an
object opposes an electric current through it. The SI unit of electrical
resistance is the ohm. Its reciprocal quantity is electrical conductance measured in siemens.
Electrical resistance shares some conceptual parallels with the mechanical
notion of friction.
The resistance of an object
determines the amount of current through the object for a given voltage across the
object.
![]()
where
R is the resistance of the object, measured in ohms,
equivalent to J·s/C2
V is the voltage across the object, measured in volts
I is the current through the object, measured in amperes
For a wide variety of materials
and conditions, the electrical resistance does not depend on the amount of
current through or the amount of voltage across the object, meaning that the
resistance R is constant.
Resistive loss
When there is a current I
through an object with resistance, R, electrical energy is converted
to heat at a rate (power) equal to
![]()
where
P is the power measured in watts
I is the current measured in amperes
R is the resistance measured in ohms
This energy conversion is useful
in applications such as incandescent lighting and electric
heating but is considered a loss in other applications such as power
transmission. Ideally, the conductors used to connect electrical devices
together should have zero resistance but in reality, only superconductors
achieve this ideal. Common ways to combat resistive loss in conductors include
using thicker wire and higher voltages.
Resistance of a conductor
DC resistance
As long as the current
density is totally uniform in the conductor, the DC
resistance R of a conductor of regular cross section can be computed as
![]()
where
l is the length of the conductor, measured in meters
A is the cross-sectional area, measured in square
meters
ρ (Greek: rho) is the electrical resistivity (also called specific
electrical resistance) of the material, measured in ohm · meter.
Resistivity is a measure of the material's ability to oppose the flow of
electric current.
For practical reasons, almost any
connections to a real conductor will almost certainly mean the current density is
not totally uniform. However, this formula still provides a good approximation
for long thin conductors such as wires.
AC resistance
If a wire conducts high-frequency
alternating current then the effective cross sectional area of the wire is
reduced because of the skin effect. This causes the wire resistance to
increase at a rate of 10dB/decade for wire radius much greater than skin depth.
In a conductor close to others,
the actual resistance is higher than that predicted by the skin effect because
of the proximity effect.
Causes of resistance
In metals
A metal consists of a
lattice of atoms,
each with a shell of electrons. This can also be known as a positive ionic
lattice. The outer electrons are free to dissociate from their parent atoms and
travel through the lattice, creating a 'sea' of electrons, making the metal a
conductor. When an electrical potential difference (a voltage) is
applied across the metal, the electrons drift from one end of the conductor to
the other under the influence of the electric
field.
Near room temperatures, the
thermal motion of ions is the primary source of scattering of electrons (due to
destructive interference of free electron wave on non-correlating potentials of
ions) - thus the prime cause of metal resistance. Imperfections of lattice also
contribute into resistance, although their contribution in pure metals is
negligible.
The larger the cross-sectional
area of the conductor, the more electrons are available to carry the current,
so the lower the resistance. The longer the conductor, the more scattering
events occur in each electron's path through the material, so the higher the
resistance. And different materials also affect the resistance.
In semiconductors and insulators
In metals, the fermi level lies
in the conduction band giving rise to free conduction electrons. However, in
semiconductors the position of the fermi level is within the band gap, exactly
half way between the conduction band minimum and valence band maximum for
intrinsic (undoped) semiconductors. This means that at 0 Kelvin, there are no
free conduction electrons and the resistance is infinite. However, the resistance
will continue to decrease as the charge carrier density in the conduction band
increases. In extrinsic (doped) semiconductors, dopant atoms increase the
majority charge carrier by donating electrons to the conduction band or
accepting holes in the valence band. For both types of donor or acceptor atoms,
increasing the dopant density leads to a reduction in the resistance. Highly
doped semiconductors hence behave metallic. At very high temperatures, the
contribution of thermally generated carriers will dominate over the
contribution from dopant atoms and the resistance will decrease exponentially
with temperature.
In ionic liquids/electrolytes
In electrolytes,
electrical conduction happens not by band
electrons or holes, but by full atomic species (ions) traveling, each
carrying an electrical charge. The resistivity of ionic liquids varies
tremendously by the salt concentration - while distilled water is almost an
insulator, salt water is a very efficient electrical conductor. In biological
membranes, currents are carried by ionic salts. Small holes in the
membranes, called ion channels, are selective to specific ions and
determine the membrane resistance.
Resistance of various materials
|
Material |
Resistivity, ρ |
|
10 - 8 |
|
|
variable |
|
|
variable |
|
|
1016 |
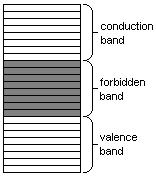
Band theory
Electron energy
levels in an insulator.
Quantum mechanics states that the
energy of an electron in an atom cannot be any arbitrary value. Rather, there
are fixed energy levels which the electrons can occupy, and values in between
these levels are impossible. The energy levels are grouped into two bands: the valence
band and the conduction band (the latter is generally above the
former). Electrons in the conduction band may move freely throughout the
substance in the presence of an electrical field.
In insulators and semiconductors,
the atoms in the substance influence each other so that between the valence
band and the conduction band there exists a forbidden band of energy levels,
which the electrons cannot occupy. In order for a current to flow, a relatively
large amount of energy must be furnished to an electron for it to leap across
this forbidden gap and into the conduction band. Thus, even large voltages can
yield relatively small currents.
Differential resistance
When resistance may depend on
voltage and current, differential resistance, incremental resistance
or slope resistance is defined as the slope of the U-I graph at a
particular point, thus:
![]()
This quantity is sometimes called
simply resistance, although the two definitions are equivalent only for
an ohmic component such as an ideal resistor. If the U-I graph is not
monotonic (i.e. it has a peak or a trough), the differential resistance will be
negative for some values of voltage and current. This property is often known
as negative resistance, although it is more
correctly called negative differential resistance, since the absolute
resistance U/I is still positive.
Temperature-dependence
Near room temperature, the electric
resistance of a typical metal varies linearly with the temperature.
At lower temperatures (less than the Debye
temperature), the resistance decreases as T5 due to the
electrons scattering off of phonons. At even lower temperatures, the dominant scattering
mechanism for electrons is other electrons, and the resistance decreases as T².
At some point, the impurities in the metal will dominate the behavior of the
electrical resistance which causes it to saturate to a constant value.
Matthiessen's Rule says that all of these different behaviors can be summed up
to get the total resistance as a function of temperature,
![]()
where Rimp is
the temperature independent electrical resistivity due to impurities, and a,
b, and c are coefficients which depend upon the metal's
properties.
The electric resistance of a
typical intrinsic (non doped) semiconductor
decreases exponentially with the temperature:
![]()
Extrinsic (doped) semiconductors
have a far more complicated temperature profile. As temperature increased starting
from absolute zero they first decrease steeply in resistance as the carriers
leave the donors or acceptors. After most of the donors or acceptors have lost
their carriers the resistance starts to increase again slightly due to the
reducing mobility of carriers (much as in a metal). At higher temperatures it
will behave like intrinsic semiconductors as the carriers from the
donors/acceptors become insignificant compared to the thermally generated
carriers.
The electric resistance of
electrolytes and insulators is highly nonlinear, and case by case dependent,
therefore no generalized equations are given.
Measuring resistance
An instrument for measuring
resistance is called a resistance meter or ohmmeter.
Simple ohmmeters cannot measure low resistances accurately because the
resistance of their measuring leads causes a voltage drop that interferes with
the measurement, so more accurate devices use four-terminal sensing.
References
- A. Matthiessen, Rep. Brit.
Ass. 32, 144 (1862)
- A. Matthiessen, Progg.
Anallen, 122, 47 (1864)